Contact lens drug delivery
Current status, technologies, and future directions
Contact lenses (CLs) have evolved from simple refractive devices into sophisticated biomedical platforms capable of a wide variety of functions, including diagnosing systemic and ocular disease (figure 1), monitoring physiology, being used for a number of advanced optical functions (including text magnification and zooming for low vision sufferers, object recognition and augmented reality) and, the purpose of this review, delivering topical ophthalmic drugs.1-18
While topical eye drops remain the dominant mode of therapy for anterior segment disease, they suffer from major shortcomings19-21 Less than 5% of an instilled drug ultimately reaches the intended ocular tissues, due to reflex tearing, blinking, rapid nasolacrimal drainage, and systemic absorption through the conjunctival blood vessels.11, 19-21 In older patients, rheumatoid arthritis affecting the hands and shoulders, poor dexterity, tremor, reduced grip strength and poor aim may make topical drop instillation difficult. In addition, noncompliance with drug instillation (which exceeds 50% of users with long term administration of drugs such as those used to treat glaucoma) further diminishes treatment efficacy. These issues result in patients frequently receiving subtherapeutic doses and experiencing high variability in exposure to the active drug.19
By placing the drug-laden reservoir directly against the ocular surface, a CL can sustain drug release for extended periods and significantly increase bioavailability. This concept was originally proposed by Wichterle in a 1965 patent on polyHEMA, the first ever soft lens material, where he speculated that “medicinal active substances such as antibiotics may be dissolved in the aqueous constituent of the hydrogels to provide medication over an extended period”.22 In the ensuing 60 years, where has the CL industry come with respect to this concept of using soft lenses for delivery of topical ocular drugs?
Growing interest
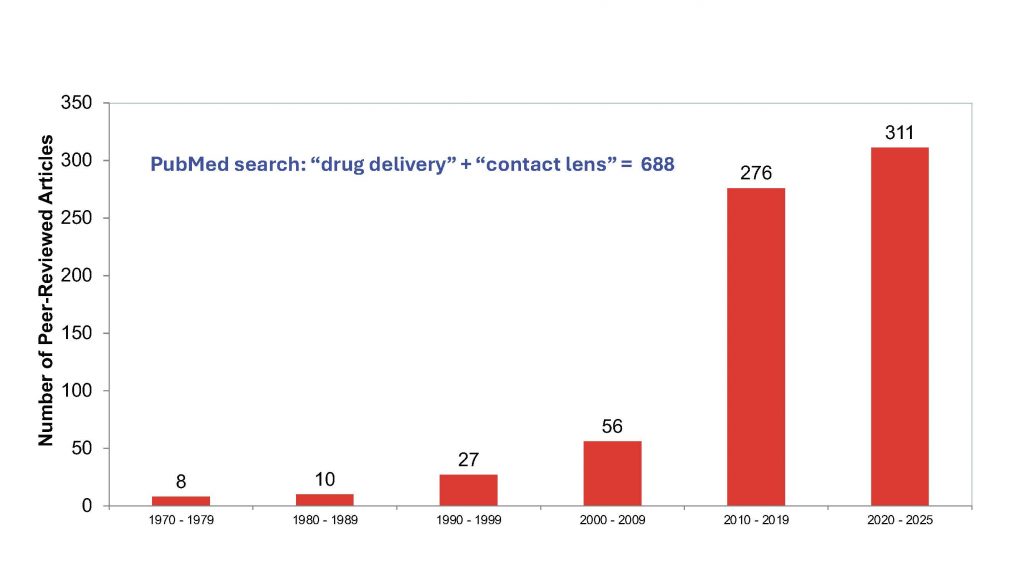
The limitations of eyedrop-based therapy have certainly resulted in growing interest in the potential for CL–based drug delivery, as shown in figure 2. In the 39 years between 1970 and 2009, only 101 peer-reviewed articles on this topic were published. In the last six years alone, over 300 articles on this topic were published, representing 45% of the total articles on this concept ever published. One of the major areas of interest relate to technologies that can control both the uptake, and of greater relevance, the controlled release of the drug of interest from the lens material. Ideally, currently available commercial CL materials would be used, and many of these have been studied for their potential use.23 However, these materials have proven to be incapable of delivering drugs to the ocular surface for a long enough time.11, 12, 23
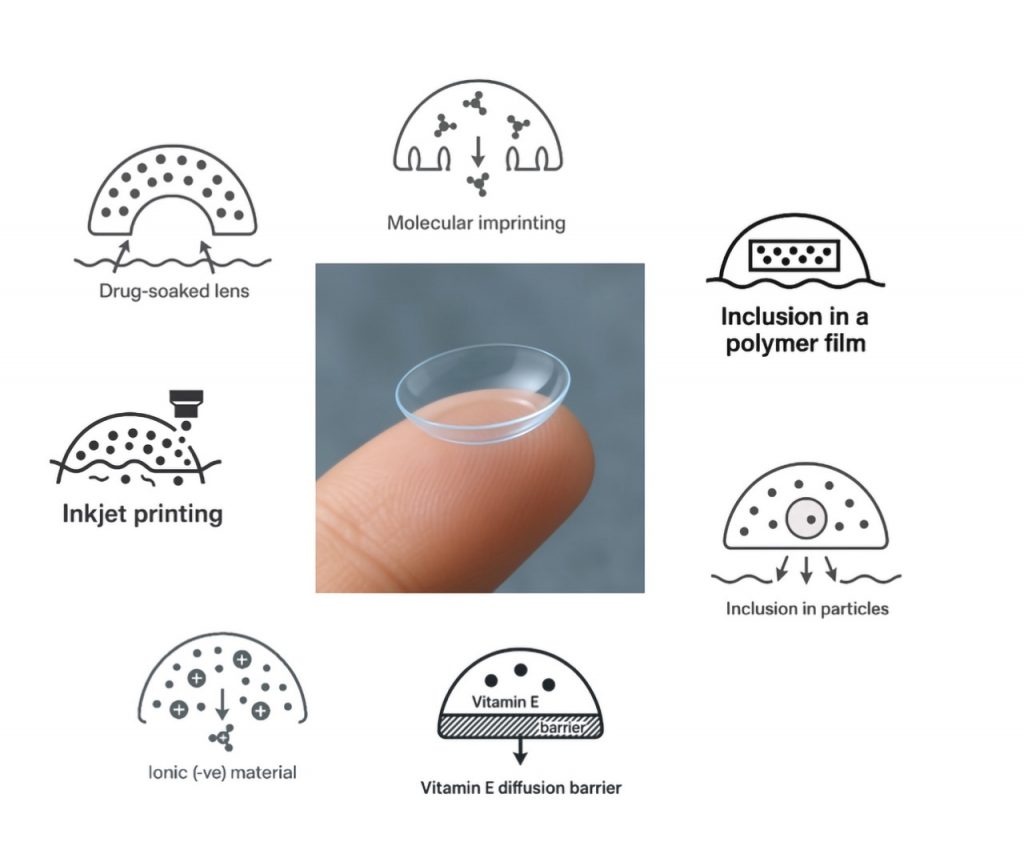
Figure 3 graphically describes the most common strategies used with drug-eluting CL to slow drug release, each rooted in classical pharmacokinetic principles.11, 20, 23-28
1. Matrix diffusion systems
In the simplest configuration, drugs are merely dissolved within currently available soft lenses and release occurs as the drug diffuses out of the polymer matrix. This mechanism underlies an early “soaked lens” approach, but this concept suffers from overly rapid release, as demonstrated by ciprofloxacin release curves from commercial hydrogels versus silicone hydrogels.29, 30 The data shows that hydrogel lenses release more drug than silicone hydrogels, and that the majority of the release occurs in just a few hours, preventing such a concept being a viable option for management of topical ocular disease.
2. Molecular imprinting
This is among the most studied technologies. Monomers are polymerized in the presence of a target molecule (the template), which is removed post polymerization, creating specific binding “pockets” or cavities for the drug of interest. This significantly increases the affinity of the drug for the CL polymer, slowing diffusion from the lens.22, 31 In a microbial keratitis rabbit model, ciprofloxacin-eluting imprinted lenses reduced bacterial burden more effectively than hourly drop therapy, illustrating the translational potential of imprinted systems.32
3. Inclusion in a polymer film
Here, a drug-loaded core is surrounded by a diffusion-limiting membrane (e.g., a PLGA film). A pHEMA-PLGA “sandwich lens” released ciprofloxacin over several weeks,33 and later iterations delivered latanoprost effectively in glaucoma-model primates, a major advance showing equivalence to daily topical therapy.34
4. Inclusion in particles
Drug-loaded nanoparticles, liposomes and micelles have been incorporated into various CL polymers and can significantly slow the release of topical ophthalmic drugs.35-41
5. Vitamin-E diffusion barriers
Vitamin E forms hydrophobic nanoclusters within the polymeric matrix, increasing tortuosity and significantly slowing drug diffusion of numerous topical drugs, as demonstrated in both laboratory and animal studies, including those using commercially available materials.22, 42-44
6. Ionic interactions
Using electrostatic interactions, positively charged drugs (e.g., ketotifen) can bind to negatively charged (ionic) commercial hydrogel matrices.45, 46 This is the approach used by Johnson & Johnson to develop the (now discontinued) Acuvue TheraVision lens, which is the only drug delivering CL to-date to be commercialised. The lens was approved for release in Japan and Canada in 2021 and the U.S. in 2022, and discontinued in 2024 due to manufacturing, sales, and regulatory concerns. Each daily disposable lens contained ketotifen, an H1 histamine receptor antagonist that stabilizes mast cells, the same active ingredient found in over-the-counter allergy eye drops like Zaditor (Alcon) and Alaway (Bausch & Lomb). The lens was designed to slowly release the drug into the eye, providing itch prevention within minutes, that was sustained for up to twelve hours after insertion.47
7. Surface printed systems
Inkjet-printed drug layers have the potential to integrate well with modern lens fabrication processes and supports precise, spatially controlled dosing and non-invasive verification of drug quantity, a major regulatory advantage.26
| Disease | Examples of drugs studied for CL delivery |
|---|---|
| Anti-allergy (figure 4) | Epinastine; Ketotifen; Olopatadine |
| Anti-infectives | Antibiotics (ciprofloxacin; gentamicin; moxifloxacin; tobramycin) Antifungals (fluconazole; natamycin) Anti-amoebic compounds Antivirals (acyclovir) |
| Corneal abrasions | Antibiotics (ciprofloxacin; moxifloxacin) NSAIDs (bromfenac; diclofenac; ketorolac) Steroid (dexamethasone) |
| Dry eye | Cyclosporine; Diquafosol; Resveratrol |
| Glaucoma | Bimatoprost; Brimonidine; Latanaprost; Timolol |
| Myopia | Atropine |
| Post surgery (cataract and refractive) | NSAIDs; steroids; antibiotics (see above for examples) |
Table 1: Diseases that may benefit from management with a CL-drug delivery device.
Given the growing interest in CL-based drug delivery systems, what diseases may be targeted for this concept? These diseases can be broadly broken down into two categories – those that require short term management (such as corneal infections and abrasions) and those requiring longer-term management over years (such as allergy, glaucoma, and dry eye). Table 1 describes a variety of ocular diseases that have been identified as potentially being served by this idea and studies published on their potential use.
Despite strong clinical and scientific rationale, commercial success has been very slow and significant manufacturing, safety, and regulatory challenges remain:
- Manufacturing complexity: Even minor leakage of the drug into the blister-pack solutions may degrade shelf-life of the product. Uniform drug loading requires precise quality control during the manufacturing process and some promising technologies (molecular imprinting, nanoparticles) are not yet optimized for mass production.
- Regulatory classification: Drug-eluting CLs are classified as “combination products”, requiring both device and pharmaceutical approvals. This complicates regulatory trial design, ethics approvals, labeling, and the regulatory pathway to commercialisation.
- Ethical approvals: While an antibiotic delivering CL has attracted much attention and many publications, obtaining ethics approval for human clinical trials for such a product is fraught with complications.
- Development costs, adoption, and market dynamics: The discontinuation of both a photochromic CL (Acuvue OASYS with Transitions) and the previously described antihistamine-releasing CL in 2024 emphasizes the impact of manufacturing costs, reimbursement challenges, regulatory barriers, and market education gaps on the commercial viability of unique CL-based technologies.
- Ethical, clinical, and prescriptive considerations: A further consideration relates to who will be licensed to prescribe these devices; optometrists, ophthalmologists, both? New clinical guidelines and prescriber education will be critical to their broad adoption.
- In Vivo vs In Vitro correlation: Predicting human pharmacokinetics remains a challenge, necessitating advanced simulators to improve translational accuracy.48-51
In closing, CL-based ocular drug delivery has transitioned from an aspirational concept to a clinically validated and technologically diverse field. Although therapeutic lenses currently represent a small fraction of commercial CL products, the scientific foundation is strong, with multiple validated delivery mechanisms, successful clinical trials, and at least one commercialized lens demonstrating real-world benefit. However, major hurdles remain, especially in manufacturing scalability, regulatory approval, and market adoption, but the convergence of biomaterials, bioelectronics, advanced printing, and AI-enabled analytics positions drug-delivering lenses as a transformative technology. The future of CL is clearly not limited to vision correction; it encompasses diagnosis, monitoring, comfort enhancement, and targeted drug therapy – ushering in a new era of personalized ophthalmic care.
Dr. Lyndon Jones is a professor at the School of Optometry & Vision Science and principal scientist at the Centre for Ocular Research & Education (CORE) at the University of Waterloo. He holds an optometry degree from the University of Wales and a PhD in chemical engineering from Aston University. His research focuses on contact lens materials, dry eye, and ocular drug delivery. He has published over 350 peer-reviewed papers and delivered more than 1200 lectures worldwide.
References:
- Jones L, Hui A, et al.: CLEAR – Kontaktlinsentechnologien der Zukunft. Cont Lens Anterior Eye 2021; 44;2: 398–430.
- Wu KY, Dave A, et al.: Intelligente Kontaktlinsen in der Augenheilkunde: Innovationen, Anwendungen und Zukunftsaussichten. Micromachines (Basel) 2024; 15;7.
- Liu X, Ye Y, et al.: Intelligente Kontaktlinsen für die Gesundheitsüberwachung und -therapie. ACS Nano 2024; 18;9: 6817–6844.
- Takaki Y: Die Zukunft der AR-Anzeige: Holografische Kontaktlinsen. JSAP Review 2023; 2023.
- Gao D, Yan C, et al.: Medikamentenfreisetzende Kontaktlinsen: Fortschritte, Herausforderungen und Perspektiven.Biointerphases 2024; 19;4.
- Shaker LM, Al-Amiery A, et al.: Die Zukunft des Sehens: Ein Überblick über die Technologie elektronischer Kontaktlinsen.ACS Photonics 2023.
- Lovrec-Krstic T, Orthaber K, et al.: Review of Potential Drug-Eluting Contact Lens Technologies. Materials (Basel) 2023; 16;10.
- Kazanskiy NL, Khonina SN, et al.: Intelligente Kontaktlinsen – ein Schritt in Richtung nicht-invasiver kontinuierlicher Überwachung der Augengesundheit. Biosensors (Basel) 2023; 13;10.
- Shi Y, Jiang N, et al.: Ophthalmologische Sensortechnologien für die Diagnose von Augenerkrankungen. Analyst 2021; 146;21: 6416–6444.
- Yang H, Zhao M, et al.: Kontaktlinsen als neue Plattform für die Verabreichung von Augenmedikamenten: Eine systematische Übersicht. Asian J Pharm Sci 2023; 18;5: 100847.
- Franco P, De Marco I: Kontaktlinsen als ophthalmologische Arzneimittelabgabesysteme: Eine Übersicht. Polymers (Basel) 2021; 13;7.
- Hui A: Kontaktlinsen für die Verabreichung von Augenmedikamenten..Clin Exp Optom 2017; 100;5: 494–512.
- Rykowska I, Nowak I, et al.: Weiche Kontaktlinsen als Arzneimittelabgabesysteme: Eine Übersicht. Molecules 2021; 26;18.
- Tashima T: Ocular Drug Delivery into the Eyes Using Drug-Releasing Soft Contact Lens, in Future Pharmacology. 2024. S. 336–351.
- Toffoletto N, Saramago B, et al.: Therapeutische Augenlinsen: Eine Übersicht. Pharmaceutics 2020; 13;1.
- ., et al.: Jüngste Fortschritte bei intelligenten Materialien für die Verabreichung von Medikamenten in das Auge, in: Adv Drug Deliv Rev 2023; 200 115006.
- Wuchte LD, DiPasquale SA, et al.: In-vivo-Arzneimittelabgabe über Kontaktlinsen: Der aktuelle Stand der Forschung von den Anfängen bis heute. Journal of Drug Delivery Science and Technology 2021; 63 102413.
- Zhao L, Song J, et al.: Therapeutische Anwendungen von Kontaktlinsen-basierten Arzneimittelabgabesystemen bei Augenerkrankungen. Drug Deliv 2023; 30;1: 2219419.
- Jünemann A, Chorągiewicz T, et al.: Bioverfügbarkeit von topisch applizierten Augentropfen. Spielt die Tropfengröße eine Rolle? Ophthalmol J 2016; 1;1: 29–35.
- ., et al.: Herausforderungen bei der Kommerzialisierung von medikamentenfreisetzenden Kontaktlinsen. Journal of Drug2020; 17;8: 1133–1149.
- Lanier OL, Manfre MG, et al.: Überblick über Ansätze zur Erhöhung der ophthalmologischen Bioverfügbarkeit von Augentropfenformulierungen. AAPS PharmSciTech 2021; 22;3: 107.
- Gonzalez-Chomon C, Concheiro A, et al.: Weiche Kontaktlinsen für die kontrollierte Verabreichung von Medikamenten am Auge: 50 Jahre Entwicklung. Ther Deliv 2013; 4;9: 1141–61.
- Fan X, Torres-Luna C, et al.: Bewertung kommerzieller weicher Kontaktlinsen für die Verabreichung von Medikamenten über die Augen: Eine Übersicht. Acta Biomater 2020; 115 60–74.
- Abdi B, Mofidfar M, et al.: Therapeutische Kontaktlinsen zur Behandlung von Erkrankungen der Hornhaut und der Augenoberfläche: Fortschritte bei der verlängerten und gezielten Wirkstoffabgabe. Int J Pharm 2023; 638 122740.
- Dixon P, Shafor C, et al.: Therapeutische Kontaktlinsen: eine Patentübersicht. Expert Opin Ther Pat 2015; 25;10: 1117–29.
- Pollard TD, Seoane-Viano I, et al.: Tintenstrahldruck von Medikamenten auf Kontaktlinsen: Optimierung der Ablagerung und nicht-invasive Dosisüberprüfung. Int J Pharm X 2023; 5 100150.
- Maulvi FA, Soni TG, et al.: Eine Übersicht über therapeutische Kontaktlinsen für die Verabreichung von Medikamenten am Auge. Drug Deliv 2016; 23;8: 3017–3026.
- Nguyen DCT, Dowling J, et al.: Mit Arzneimitteln beladene Kontaktlinsen als System zur Verabreichung von Augenmedikamenten: Eine Übersicht über wichtige Methoden zur Charakterisierung von Linsen unter Bezugnahme auf ISO-Normen. Cont Lens Anterior Eye 2021; 44;6: 101487.
- Karlgard CC, Jones LW, et al.: Wechselwirkung von Ciprofloxacin mit Kontaktlinsen auf Silikonbasis und herkömmlichen Hydrogel-Kontaktlinsen. Eye Contact Lens 2003; 29;2: 83–9.
- Hui A, Boone A, et al.: Aufnahme und Freisetzung von Ciprofloxacin-HCl aus herkömmlichen und Silikon-Hydrogel-Kontaktlinsenmaterialien. Eye Contact Lens 2008; 34;5: 266–71.
- White CJ, Byrne ME: Molekular geprägte therapeutische Kontaktlinsen. Expert Opin Drug Deliv 2010; 7;6: 765–80.
- Hui A, Willcox M, et al.: In-vitro- und In-vivo-Bewertung neuartiger Ciprofloxacin-freisetzender Silikon-Hydrogel-Kontaktlinsen..Invest Ophthalmol Vis Sci 2014; 55;8: 4896–904.
- Ciolino JB, Hoare TR, et al.: Eine medikamentenfreisetzende Kontaktlinse..Invest Ophthalmol Vis Sci 2009; 50;7: 3346–52.
- Ciolino JB, Ross AE, et al.: Latanoprost-freisetzende Kontaktlinsen bei Affen mit Glaukom. Ophthalmology 2016; 123;10: 2085–92.
- Maulvi FA, Mangukiya MA, et al.: Verlängerte Freisetzung von Ketotifen aus mit Silica-Nanopartikeln beladenen Hydrogel-Kontaktlinsen: In-vitro- und In-vivo-Bewertung..J Mater Sci Mater Med 2016; 27;6: 113.
- Lu C, Mikhail AS, et al.: Hydrogele mit vernetzten Blockcopolymer-Mizellen im Kern. J Biomater Sci Polym Ed 2011.
- Patel A, Cholkar K, et al.: Okuläre Arzneimittelabgabesysteme: Ein Überblick. World J Pharmacol 2013; 2;2: 47–64.
- Gulsen D, Li CC, et al.: Dispersion von DMPC-Liposomen in Kontaktlinsen für die ophthalmologische Arzneimittelabgabe..Curr Eye Res 2005; 30;12: 1071–80.
- Liu L-C, Chen Y-H, et al.: Überblick über die jüngsten Fortschritte in der nanobasierten Augenmedikamentenverabreichung, in International Journal of Molecular Sciences. 2023.
- Maulvi FA, Patil RJ, et al.: Wirkung von Goldnanopartikeln auf die Timololaufnahme und deren Freisetzungskinetik aus Kontaktlinsen: In-vitro- und In-vivo-Bewertung. Acta Biomater 2019; 86 350–362.
- Maulvi FA, Desai DT, et al.: Fortschritte und Herausforderungen bei Kontaktlinsen mit Nanopartikeln für die Verabreichung von Augenmedikamenten. Int J Pharm 2021; 608 121090.
- Peng CC, Kim J, et al.: Verlängerte Abgabe hydrophiler Medikamente aus Silikon-Hydrogel-Kontaktlinsen mit Vitamin-E-Diffusionsbarrieren. Biomaterials 2010; 31;14: 4032–47.
- Paradiso P, Serro AP, et al.: Kontrollierte Freisetzung von Antibiotika aus mit Vitamin E beladenen Silikon-Hydrogel-Kontaktlinsen. J Pharm Sci 2016; 105;3: 1164–72.
- Dixon P, Fentzke RC, et al.: In-vitro-Arzneimittelfreisetzung und In-vivo-Sicherheit von mit Vitamin E und Cysteamin beladenen Kontaktlinsen. Int J Pharm 2018; 544;2: 380–391.
- Bengani LC, Chauhan A: Verlängerte Freisetzung eines anionischen Wirkstoffs durch Kontaktlinsen, die mit einem kationischen Tensid beladen sind. Biomaterials 2013; 34;11: 2814–21.
- Soluri A, Hui A, et al.: Abgabe von Ketotifenfumarat durch handelsübliche Kontaktlinsenmaterialien..Optom Vis Sci 2012; 89;8: 1140–9.
- Pall B, Gomes P, et al.: Behandlung von juckenden Augenallergien mit einer Antihistamin-freisetzenden Kontaktlinse.Cornea 2019; 38;6: 713–717.
- Mohammadi S, Eslami S, et al.: In-vitro-Tränenersatzsystem: Bewertung der Wirkstoffabgabe aus Kontaktlinsen-Biomaterialien durch ein- und mehrschichtige Hornhautepithelien unter Ersatzbedingungen. Drug Deliv Transl Res 2025; 15;7: 2509–2521.
- Pereira-da-Mota AF, Vivero-Lopez M, et al.: In-vitro-in-vivo-Korrelation von Wirkstofffreisetzungsprofilen aus medikamentösen Kontaktlinsen unter Verwendung eines In-vitro-Augenblinzelmodells. Drug Deliv Transl Res 2023; 13;4: 1116–1127.
- Pereira-da-Mota AF, Phan CM, et al.: Testen der Wirkstofffreisetzung aus medikamentösen Kontaktlinsen: Das fehlende Glied zur Vorhersage der In-vivo-Leistung..J Control Release 2022; 343 672–702.
- Phan CM, Shukla M, et al.: Entwicklung eines In-vitro-Blinzelmodells für die Verabreichung von Augenmedikamenten.Pharmaceutics 2021; 13;3.